The Changing Face of Shark and
Ray Classification
Elasmobranch classification ain't what it used to be. For roughly 2,000 years, elasmobranch classification was deceptively straight-forward: if it was torpedo-shaped, it was a shark or dogfish; if pancake-shaped, a skate or ray; if it had six or more pairs of gill slits it was 'primitive'; if five pairs of gill slits, it was 'advanced'. But the days are long gone when sharks and rays can be classified according to easily observable, external characteristics.
By the 1850's, it was clear that this simplistic approach was inadequate to reflect the evolutionary and structural diversity of these cartilaginous fishes. Over the next 120 years or so, numerous elasmobranch classificatory schemes were proposed, each achieving various degrees of success. Then, in 1973, systematist Leonard J.V. Compagno proposed a pivotal scheme for classifying all extant elasmobranchs. Based on his extensive comparative studies of elasmobranch crania, jaw and gill arches, teeth, muscles, vertebrae and other skeletal elements, Compagno divided these fishes into thirteen orders of unequal size but attractively uniform characteristics. For the first time, ichthyologists had a classificatory scheme that adequately reflected elasmobranch diversity in a manageable number of what seemed to be truly 'natural' groups. For two-and-a-half decades, Compagno's scheme reigned supreme and serene conformity presided over elasmobranch classification.
Alas, this halcyon state of affairs was not to remain. With the advance of new systematic paradigms and the development of powerful new tools (most notably, molecular genetics), elasmobranch classification has undergone some major revisions in recent years. Systematic up-starts - such as Marcelo de Carvalho, Guido Dingerkus, Peter Last, Andrew P. Martin, John McEachran, Brian Mould, Gavin J.P. Naylor, Joseph Nelson, Kiyonori Nishida, and Shigeru Shirai - have all proposed minor-to-radical modifications to Compagno's basic scheme. It will be many years before the merits of the schemes proposed by each of these respective workers can be properly assessed. What follows is thus a provisional synthesis of what seems to me the most compelling of current ideas about elasmobranch classification. I am sure there are those who would favor a classificatory scheme different from the one I use here. But - even if part or all of my proposed scheme is eventually shown to be incorrect or downright naive - such debate only advances the science of elasmobranch systematics.
Cladograms:
Picturing Evolutionary Relationships
Interrelationships among the various elasmobranch orders can be represented in a branching diagram called a "cladogram". Each bifurcating branch on a cladogram indicates an evolutionary split, or divergence, from a common ancestor. Therefore, the fewer bifurcations separating groups of organisms (identified on the right side of the cladogram), the more recently they shared a common ancestor and the more closely they are related.
Reading a cladogram is wonderfully intuitive: they are read from left to right, like any English-language sentence. The base of the horizontal 'trunk' at the extreme left represents the common ancestor of the entire taxon considered. The common ancestor is defined by a suite of characters shared (in some fashion) by all its descendants. Descendants are represented by 'branches' arising to the right of the horizontal 'trunk'. A vertical 'branch' represents an evolutionary divergence, the point in a lineage where new derived characters first appear. Each divergence - or, more formally, branching point - is termed a "node". Individual nodes are defined by a suite of shared derived characters (synapomorphies).
Characters become increasingly specific as one progresses from left to right, through further divergences, to subsequent nodes. The tips of the horizontal 'limbs' to the right represent the individual taxa listed beside them. The closer together any two 'limbs', the more closely-related the groups they represent. Any pair or cluster of horizontal 'limbs' that can be traced back to a common vertical 'branch' share a common ancestor and are termed "sister groups". The fewer bifurcations between sister groups, the more recent their common ancestor and the more closely related they are. Character suites are typically identified by numbers marked on or near each node of the cladogram; the individual characters defining each node (and thus used to construct the cladogram) are usually listed below the diagram, but we won't bother with doing so here.
The cladogram accompanying the synopsis of each elasmobranch order or group throughout this website thus graphically represents the most likely pattern of evolutionary divergence of extant sharks and rays from their common ancestor. The group being discussed is highlighted by being placed in a box, indicating its phylogenetic position among other groups. The system adopted here divides elasmobranchs into eleven groups (mostly considered at the order level), with the batoids lumped into a single unit for brevity and convenience.
Galea and Squalea:
Two Great Branches of Elasmobranch Evolution
Most lineages of extant elasmobranchs originated during the early Jurassic and late Cretaceous periods (between about 200 and 100 million years ago). Most of these lineages seem to have been evolving along separate paths ever since but not all lineages represented in the fossil record left living descendants, making it difficult to sort out the phylogeny and classification of extant sharks and rays. Despite these difficulties, living elasmobranchs fall readily into two groups, the Galea and the Squalea. The squaleans constitute about two-thirds of living elasmobranchs and are generally regarded as less 'advanced' than the galeans, although it can be argued that the squalean group is at least as derived as the most 'advanced' galean sharks. The sharks comprising the Galea (traditionally referred to as the superorder Galeomorphii) are the bullheads (Heterodontiformes), carpet sharks (Orectolobiformes), ground sharks (Carcharhiniformes), and - the group that includes the great white - the mackerel sharks (Lamniformes) Of these, the heterodontoids are regarded as the most ancestral, and the carcharhinoids the most derived.
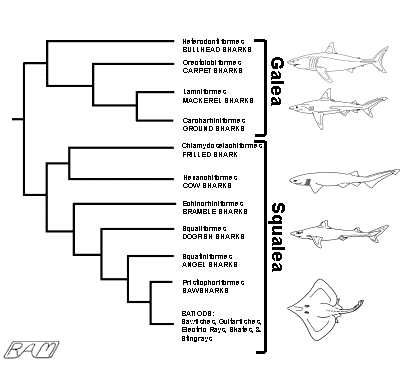 From
the accompanying cladogram, we can see that the Great White and other lamniform
sharks are most closely related to the carcharhinoids. It is not known exactly
when the common ancestor of the lamnoids and carcharhinoids diverged, nor what
this common ancestor might have looked like. The fossil record - scattered and
incomplete as it is - seems to suggest that the carcharhinoids and lamnoids may
have shared a common ancestor in the late Jurassic Period, about 150 million
years ago. At that time, the world was uniformly warmer than it is now and there
was only one sea, Panthalassia, surrounding the single supercontinent, Pangaea.
As Pangaea broke apart into discrete continents and the climate cooled, the
carcharhinoids somehow retained their stronghold in warm coastal waters and -
although a few species have successfully invaded cool waters - they are still
most richly represented in warm temperate and tropical seas. Although the
lamnoids continued to be represented in warm waters, at some stage in their
evolution they became predominantly creatures of cooler waters - a few species
haunting the chill dark depths, others making a living in cool temperate and
boreal seas.
From
the accompanying cladogram, we can see that the Great White and other lamniform
sharks are most closely related to the carcharhinoids. It is not known exactly
when the common ancestor of the lamnoids and carcharhinoids diverged, nor what
this common ancestor might have looked like. The fossil record - scattered and
incomplete as it is - seems to suggest that the carcharhinoids and lamnoids may
have shared a common ancestor in the late Jurassic Period, about 150 million
years ago. At that time, the world was uniformly warmer than it is now and there
was only one sea, Panthalassia, surrounding the single supercontinent, Pangaea.
As Pangaea broke apart into discrete continents and the climate cooled, the
carcharhinoids somehow retained their stronghold in warm coastal waters and -
although a few species have successfully invaded cool waters - they are still
most richly represented in warm temperate and tropical seas. Although the
lamnoids continued to be represented in warm waters, at some stage in their
evolution they became predominantly creatures of cooler waters - a few species
haunting the chill dark depths, others making a living in cool temperate and
boreal seas.
In this way, these two 'sister groups' crudely divided the resources of the world ocean: the carcharhinoids exploiting mostly warm seas, the lamnoids - including the white shark - mostly cool. That these two groups of galean sharks, starting from a common origin, managed to specialize in such fundamentally different habitats is testimonial to the wondrous ecological plasticity of elasmobranchs.
Update:
Based on strong evidence of several independent molecular studies, the batoids (skates and rays) are no longer considered terminal (derived) Squaleans, but a monophyletic elasmobranch group separate from the sharks. The sharks remain divided into galeomorphs (including bullhead, mackerel, carpet, and ground sharks; equal to Galea of the previous phyletic scheme) and Squalomorphs (including cow & frilled sharks, bramble sharks, dogfishes, angel sharks, and sawsharks; similar to Squlea minus the batoids). The remainder of this site is being re-worked to reflect this new interpretation.
Although removal of the batoids from the Squlea is generally agreed upon, this phyletic modification is not without its problems. Most notably, the stability of the Squalomorph clade is greatly weakened by removal of the batoids.
Another recent change, which is so new it has not yet been supported or refuted by independent studies, is the hypothesis that heterodontoids (bullhead sharks) are not basal Galeomorphs but Squalomorph sharks closely allied with the Squaliformes (dogfishes). Hopefully, further data will clarify phyletic relationships among elasmobranchs.
