Biology of the Bluntnose Sixgill
Adapted from a PowerPoint presentation given by R. Aidan Martin at a Sixgill Shark conference held at the Vancouver Aquarium, Vancouver, Canada, in November 2000
Identification
|
Distribution
The Bluntnose Sixgill may be the most widely distributed of all sharks, rivalled only by the White Shark (Carcharodon carcharias) |
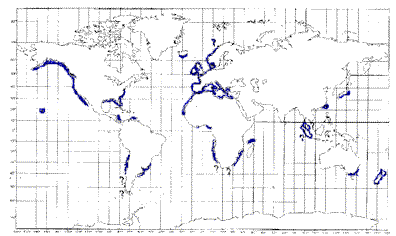 |
Habitat
- benthic on continental and insular shelves and upper slopes, from the surface and the intertidal down to a depth of at least 8,200 feet (2 500 metres)
- young often close inshore, adults typically in deeper water below 300 feet (90 metres)
- strongly bottom-associated, mostly over bathic soft bottoms and rocky reefs
- a vertical migrator (moving surfaceward at night, returning to the depths before dawn)
- seasonally (May-November) enters uncharacteristically shallow waters off British Columbia and Washington State, where it is typically encountered by divers at depths of 75 to 130 feet (23 to 39 metres) by day and as shallow as 10 feet (3 metres) at night
- one report (Clark, pers. comm.) off a deep coral reef face in Palau during daylight hours
- several local areas well known for shallow-water Sixgills
- wide range and diversity of habitats suggest they may be encountered in many locations along our coast
Diet
Molluscs
- snail (Gastropoda) South Africa
- Chokka Squid (Loligo vulgaris reynaudii) South Africa
- Angola Flying Squid (Todarodes angolensis) South Africa
Crustaceans
- crabs (Brachyura) Cuba
- shrimps (Caridia) Cuba
Agnathans
- Pacific Hagfish (Eptatretus tridentata) California and British Columbia
- sea lampreys (Petromyzontidae)
Teleost
- Cape Anchovy (Engraulis capensis) South Africa
- Pacific salmon (Oncorhynchus spp.) British Columbia
- Spotted Hake (Urophycis regia) Florida
- Luminous Hake (Steindachneria argentea) Florida
- Atlantic hakes (Merluccius spp.) South Africa and Spain
- Pacific Hake (Merluccius productus) California
- cods (Gadidae)
- lings (Gadidae)
- Lingcod (Ophiodon elongatus) British Columbia
- Cabezon (Scorpaenichthys marmoratus) British Columbia
- Jacopever (Helicolenus dactylopterus) South Africa
- rockfish (Sebastes spp.) British Columbia
- grouper (Serranidae) Florida
- grenadier (Coelorinchus sp.) South Africa
- gurnards (Triglidae)
- Dolphinfish (Coryphaena hipparus) Cuba
- Chub Mackerel (Scomber japonicus) South Africa
- Snoek (Thyrsites atun) South Africa
- small marlins (Makaira spp.) Cuba
- small Swordfish (Xiphias gladius) Azores and Cuba
- flounders (Pleuronectiformes)
- anglerfishes (Lophiformes)
Chondrichthyans
- Ratfish (Hydrolagus colliei) California and British Columbia
- Saint Joseph (Callorhinchus capensis) South Africa
- Bluntnose Sixgill Shark (Hexanchus griseus) Cuba [scavenged]
- Spiny Dogfish (Squalus acanthias) South Africa, California and British Columbia
- Longnose Dogfish (Squalus blainvillei) Hawaii
- Shortnose Dogfish (Squalus megalops) South Africa
- Prickly Shark (Echinorhinus cookei) California
- catsharks (Scyliorhinidae) South Africa
- Smoothhound Shark (Mustelus mutelus) South Africa
- other sharks South Africa and Cuba
- Atlantic Torpedo (Torpedo nobiliana) Spain
- Biscuit Skate (Raja clavata) South Africa
Marine Mammals
- Harbour Seal (Phoca vitulina richardsi) British Columbia
- Cape Fur Seal (Arctocephalus pusullus pusillus) South Africa
- pinniped remains California
- Bottlenose Dolphin (Tursiops truncatus) South Africa [scavenged]
- whale blubber California
Feeding Behavior
- multicusped lower dentition suggests gouging or sawing flesh from food items too large to be swallowed whole
- given speedy prey, probably relies on stealth to approach prey, launching a sudden ambush attack from close range (< 3 body lengths)
- may lunge with jaws gaping, catching smaller prey at a jaw corner with a lateral swipe of the head
- use of bottom topography to stalk and capture prey (i.e. Lingcod, pers obs.)
Predators
- Stellers Sea Lion (Eumetopias jubatus)?
- Killer Whale (Orcinus orca)?
- White Shark (Carcharodon carcharias)?
Ecological Competitors
- Pacific Sleeper Shark (Somniosus pacificus)?
- White Shark (Carcharodon carcharias) possible competitive exclusion in South Africa
Sensory Biology
- brain is proportionately small, with a smooth-surfaced, symmetrical cerebrum (Bauchot et al. 1995 and pers. obs.)
- olfactory bulbs very large, comprising about 8% of total brain weight (Martin, unpubl. data)
- facial and trigeminal nerves proportionately small in diameter, suggesting electroreception relatively unimportant
- nuclei of lateral-line lobes proportionately small, suggesting acoustico-lateralis system of limited importance
- epiphyseal window over pineal organ may help regulate seasonal changes in reproductive hormones
- attempts at maintenance in aquaria indicate that Bluntnose Sixgills become highly stressed even at moderate light levels; eyes well adapted to deep-sea light conditions
- retinas completely populated by rods (no cones), with long rod outer segments (averaging 26.4 mm) and peak spectral sensitivity at about 460 nm (Cohen 1985)
- iris non-contractile; tapetum lucidum present and migratory pigment cells absent (Martin, unpubl. data)
These data suggest that:
- social organization is minimal and schooling behavior is unlikely
- olfaction and vision are probably most important in prey detection and social interactions (courtship and agonistic encounters, respectively)
Reproduction
- Length at Maturity: males 11 feet (3.4 metres), females 14 feet (4.3 metres)
- Age at Maturity: unknown for both sexes (age determination hampered by poorly calcified vertebrae), but probably at least 11-14 years for males and 18 to 35 years for females
- Mode: ovoviviparity
- Gestation: unknown, but probably more than 2 years
- Number of Pups: 22-108; large litter size suggests high juvenile mortality
- Length at Birth: 28 inches (70 centimetres)
- Maximum Length: 16 feet (4.8 metres)
- Longevity: unknown, but probably more than 80 years
Social Behavior
 As in other
hexanchoids (but not chlamydoselachoids!) strong sexual dimorphism in teeth
male has more erect primary cusps than female, especially noticeable on
anteriormost cusp of enlarged lower anterior teeth of male, which is about
50% longer than the secondary cusp that follows it
As in other
hexanchoids (but not chlamydoselachoids!) strong sexual dimorphism in teeth
male has more erect primary cusps than female, especially noticeable on
anteriormost cusp of enlarged lower anterior teeth of male, which is about
50% longer than the secondary cusp that follows it
- courtship and mating never observed
- as a releaser, courting male probably gently nips females gill region, pectoral fins, and flanks
- seasonal appearance of scars on females in shallow-water
- agonistic display features strong pectoral fin depression and exaggerated jaw gapes
- has been known to snap aggressively at divers when touched
- many of the same individuals appear to return to favored shallow-water sites, year-after-year providing long-term opportunities to interact socially with one another
So What Are They Doing in Shallow Water?
My hypothesis:
- light-sensitive sharks seasonally enter uncharacteristically shallow coastal waters of Pacific Northwest under cover of plankton bloom
- arrival in highly productive shallow waters coincides with local salmon and herring runs pups are dropped in shallow coastal nursery areas offering plenty of food but relative safety from predators
- adults and subadults take advantage of rich feeding and opportunities for social interaction just seaward of nursery areas
- when water clears in winter, the light-sensitive sharks head for deeper water (>300 feet or 100 metres deep)
Bibligraphy
Backus, R.H. 1957. Notes on Western North Atlantic Sharks. Copeia, 1957(3): 246-248.
Barans-Charles, A., and F. Ulrich-Glenn. 1995. Sixgill Shark, Hexanchus griseus, Aggressive Feeding Behavior on Epibenthic Crabs. J. Elisha Mitchell Sci. Soc., 110(1): 49-52.
Bass, A.J., J.D. DAubrey, and N. Kistnasamy. 1975. Sharks of the East Coast of Southern Africa. V. The Families Hexanchidae, Chlamydoselachidae, Heterodontidae, Pristiophoridae and Squatinidae. Oceanogr. Res. Inst. Invest. Rep. 43: 8-9.
Bauchot, Roland, Roland Platel, Jean-Marc Ridet, Monique Diagne, and Cristian Delfini. 1995. Encéphalisation et Adaptations Ecobiologiques ches les Chondrichthyens. Cybium, 19(2): 153-165.
Bigelow, Henry B., and William C. Schroeder. 1948. Fishes of the Western North Atlantic, Part One: Lancelets, Cyclostomes, Sharks. Sears Foundation for Marine Research, New Haven. pp 80-87.
Branstetter, Steven, and John D. McEachran. 1986. A First Record of Odontaspis noronhai (Lamniformes: Odontaspididae) for the Western North Atlantic, with Notes on Two Uncommon Sharks from the Gulf of Mexico. Northeast Gulf Sci., 8(2): 153-160.
Cadenat, J., and J. Blanche. 1981. Requins de Méditerranée et d Atlantique (Plus Particuliérement de la Côte Occidentale d Afrique). Faune Tropicale XXI. ORSTROM, Paris. pp 20-22.
Carey, Francis G., and Eugenie Clark. 1995. Depth Telemetry From the Sixgill Shark, Hexanchus griseus, at Bermuda. Envir. Biol. Fishes, 42: 7-14.
Castro, José I. 1983. The Sharks of North American Waters. Texas A&M University Press, College Station. p 37.
Clark, Eugenie, and Emory Kristof. 1986. Sharks at 2,000 Feet. Nat. Geogr., 170(5) [November 1986]: 680-691.
Clark, E., and E. Kristof. 1990. Deep Sea Elasmobranchs Observed From Submersibles in Grand Cayman, Bermuda, and Bahamas. NOAA Tech. Rep. NMFS 90: 275-290.
Clark, Eugenie, and Emory Kristof. 1991. How Deep Do Sharks Go? Reflections on Deep Sea Sharks. Bull. Amer. Litt. Soc, 19(4)/20(1): 79-84.
Clemens, W.A., and G.V. Wilby. 1946. Fishes of the Pacific Coast of Canada. Bull. Fish. Res. Bd. Can., 68: 52.
Coad, Brian W. 1995. Encyclopedia of Canadian Fishes. Canadian Museum of Nature, Ottawa. pp 686-687.
Cohen, J. 1985. The Visual System of the Sixgill Shark, Hexanchus griseus. ASIH/AES Abstracts, University of Tennessee, Knoxville.
Compagno, Leonard J.V. 1984. Sharks of the World. FAO Species Catalogue, Vol. 4, Part 1 (Hexanchiformes to Lamniformes). United Nations Development Programme, Rome. pp 19-20.
Costa, M.E., and T.C. Borges. 1998. Shark Discard from the Southern Portuguese Coastal Fisheries. ICES, 15: 1-20.
Crow, Gerald L., Christopher G. Lowe, and Bradley M. Wetherbee. 1996. Shark Records from Longline Fishing Program in Hawaii with Comments on Pacific Ocean Distributions. Pac. Sci., 50(4): 382-392.
Ebert, David A. 1986. Aspects on the Biology of Hexanchid Sharks along the California Coast. pp 437-449 In T. Uyeno, R. Arai, T. Taniuchi, and K. Matsuura (eds.) Indo-Pacific Fish Biology: Proceedings of the Second International Conference on Indo-Pacific Fishes. Ichthyological Society of Japan, Tokyo.
Ebert, David A. 1986. Biological Aspects of the Sixgill Shark, Hexanchus griseus. Copeia, 1986(1): 131-135.
Ebert, David A. 1994. Diet of the Sixgill Shark Hexanchus griseus off Southern Africa. S. Afr. J. Mar. Sci., 14: 213-218.
Faughnan, Victor R. 1980. The National Shark-O-Pedia. Undersea Resources, Honolulu. pp 47-48.
Hart, J.L. 1973. Pacific Fishes of Canada. Fish. Res. Bd. Can. Bull. 180: 27-28.
Herald, Earl S. 1968. Size and Aggressiveness of the Sevengill Shark (Notorynchus maculatus). Copeia, 1968(2): 412-414.
Issacs, John D., and Richard A. Schwartzlose. 1975. Active Animals of the Deep-Sea Floor. Sci. Amer., 233(4) [October 1975]: 85-91.
Kabasakal, H. 1998. The First Record of the Bluntnose Six-Gill Shark [Hexanchus griseus (Bonnaterre, 1788]) in the Sea of marmara. Acta Adriat., 39(1): 67-70.
Kato, Susumu, Stuart Springer, and Mary H. Wagner. 1967. Field Guide to Eastern Pacific and Hawaiian Sharks. US Fish and Wildlife Service, Circular 271. pp 5-6.
Kemp. Noel R. 1978. Detailed Comparisons of the Dentitions of Extant Hexanchid Sharks and Tertiary Hexanchid Teeth from South Australia and Victoria (Selachii: Hexanchidae). Mem. Nat. Mus. Vict., 39: 61-83.
Last, Peter R., and John D. Stevens. 1994. Sharks and Rays of Australia. CSIRO Australia. pp 40-41.
Lineaweaver, Thomas H., III, and Richard H. Backus. 1970. The Natural History of Sharks. J.B. Lippincott Co., Philadelphia. pp 168-170.
Lynch, D.D. 1963. First Australian Record of Hexanchus griseus (Bonnaterre) 1780. The Six-Gilled Shark. Mem. Nat. Mus. Vict., 26: 259-261.
Marliave, Jeff. 1984. Six Gill Shark: a Primeval Visitor From the Abyss. Skin Diver, 33(9) [September 1984]: 40-41.
Martin, R. 1991. Sixgill Non Sequitur. DIVER Magazine, Sept. 1991: 45-46.
Martin, R. 1998. Swimming With Jurassic Shark. Scuba Diver, November-December 1998: 66-68.
Mayhew, Anne. 1988. Diving For Deep Teeth. Beautiful British Columbia, 30(2) [Summer 1988]: 18-23.
Michael, Scott W. 1993. Reef Sharks and Rays of the World. Sea Challengers, Monterey. p 34.
Miller, Bruce, and David W. Greenfield. 1965. A Juvenile Six-Gilled Shark (Hexanchus griseus) from the San Juan Islands, Washington. J. Fish. Res. Bd. Canada, 22(3): 857-859.
Nevell, Len. 1998. Bluntnose Six-Gilled Shark. Glaucus, 9(1): 28-29
ODonoghue, C.H., and L.L. Bolton. 1926. Note on the Structure of the Lymphoid Organ (Organ of Leydig) and Spleen of Hexanchus corinus. J. Anat., 61: 40-63.
ONeil, Grady. 1995. A Summary of Data Collected During Tagging Cruises For the Sixgill Shark (Hexanchus griseus), May, June, July, and September 1994. Unpubl. Rep., Dir. Res. (Biol 490) U. of Victoria, Victoria. 48 pp.
Roedel, Phil M., and Wm. Ellis Ripley. 1960. California Sharks and Rays. Calif. Dept. Nat. Res. Div. Fish & Game, Fish Bull. No. 75. p 40.
Springer, Stewart, and Richard A. Waller. 1969. Hexanchus vitulus, a New Sixgill Shark from the Bahamas. Bull. Mar. Sci., 19(1): 159-174.
Steel, Rodney. 1985. Sharks of the World. Facts on File, New York. pp 81-83.
Stefanescu, C., D. Lloris, and J. Rucabado. 1992. Deep-Living Demersal Fishes in the Catalan Sea (Western Mediterranean) Below a Depth of 1000 m. J. Nat. Hist., 26: 197-213.
Taylor, Leighton. 1993. Sharks of Hawaii: Their Biology and Cultural Significance. University of Hawaii Press, Honolulu. p 50.
Tortonese, Enrico. 1985. Gli Squali Mediterranei del Genere Hexanchus. Atti. Soc. Ital. Sci. Nat. Museo Civ. Stor. Nat. Milano, 126(3-4): 137-140.
Wallet, Tim. 1983. Shark Attack and Treatment of Victims in Southern African Waters. C. Struik Publishers, Capetown. pp 121-122.
Went, A.E.J. 1979. Two Elasmobranchs Scarce in Irish Waters. Ir. Nat. J., 19(11): 381-383.
Wheeler, Alwyne. 1969. The Fishes of the British Isles and
North-West Europe. Macmillan, London.
p 38-39.
Zama, A. 1988. Southern Distribution of the Sixgill Shark Hexanchus griseus (Bonnaterre, 1788) and the Striped Mullet Mugil cephalus Linnaeus, 1758 in Chile. Cienc. Technol. Mar. Valparaiso, 12: 137-140.
